About
Pridopidine
Pridopidine is an oral investigational drug currently in development for the treatment of Huntington’s disease (HD) and amyotrophic lateral sclerosis (ALS). It is administered in a small, easy-to-swallow capsule twice daily. Multiple clinical studies have been conducted providing important understanding about pridopidine’s safety, mechanism of action, and efficacy.
Pridopidine has not been approved by the US Food & Drug Administration or other ex-US regulatory agencies. Its safety and efficacy have not yet been determined.
Imaging studies in humans show that pridopidine enters the brain and spinal cord, where it activates a protein called the sigma-1 receptor (S1R).
The S1R is broadly expressed in the brain and central nervous system, particularly in the spinal cord, brain stem, basal ganglia, cortex, and cerebellum, where it regulates several cellular mechanisms commonly impaired in neurodegenerative diseases, such as HD and ALS.

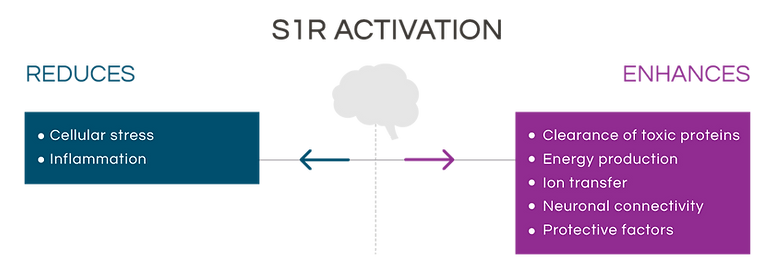
Activation of the S1R by pridopidine enhances the clearance of toxic proteins, increases energy production, and reduces cellular stress and inflammation.
These mechanisms are crucial for a neuron’s function and survival.
Studies in animal models and human cells show that pridopidine prevents neuronal cell death and can strengthen and enhance the connections between neurons.
Pridopidine has been studied in more than 1700 people, and long-term safety data are available from previous clinical studies, some running up to 7 years. In these investigational studies, pridopidine at the therapeutic dose has an observed safety and tolerability profile comparable to placebo. Most reported adverse events included falls, nausea, diarrhea, and cold-like symptoms.

Prilenia holds orphan drug designation for pridopidine in both HD and ALS in the United States and EU. In addition, pridopidine has received Fast Track designation by the U.S. Food and Drug Administration (FDA) for the treatment of HD. Drugs that receive Fast Track designation may be eligible for more frequent communications with the FDA and may also qualify for accelerated approval and priority review of new drug applications.

